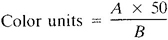
In order to promote public education and public safety, equal justice for all, a better informed citizenry, the rule of law, world trade and world peace, this legal document is hereby made available on a noncommercial basis, as it is the right of all humans to know and speak the laws that govern them.
18TH EDITION 1992
Prepared and published jointly by:
AMERICAN PUBLIC HEALTH ASSOCIATION
AMERICAN WATER WORKS ASSOCIATION
WATER ENVIRONMENT FEDERATION
Joint Editorial Board
Arnold E. Greenberg, APHA, Chairman
Lenore S. Clesceri, WEF
Andrew D. Eaton, AWWA
Managing Editor
Mary Ann H. Franson
Publication Office
American Public Health Association
1015 Fifteenth Street, NW
Washington, DC 20005
Copyright © 1917, 1920, 1923, and 1925 by
American Public Health Association
Copyright © 1933, 1936, and 1946 by
American Public Health Association
American Water Works Association
Copyright © 1955, 1960, and 1965 by
American Public Health Association
American Water Works Association
Water Pollution Control Federation
Copyright © 1971 by
American Public Health Association
American Water Works Association
Water Pollution Control Federation
Copyright © 1976 by
American Public Health Association
American Water Works Association
Water Pollution Control Federation
Copyright © 1981 by
American Public Health Association
American Water Works Association
Water Pollution Control Federation
Copyright © 1985 by
American Public Health Association
American Water Works Association
Water Pollution Control Federation
Copyright © 1989 by
American Public Health Association
American Water Works Association
Water Pollution Control Federation
Copyright © 1992 by
American Public Health Association
American Water Works Association
Water Environment Federation
All rights reserved. No part of this publication may be reproduced, graphically or electronically, including entering in storage or retrieval systems, without the prior written permission of the publishers.
30M7/92
The Library of Congress has catalogued this work as follows:
American Public Health Association.
Standard methods for the examination of water and wastewater.
ISBN 0-87553-207-1
Printed and bound in the United States of America.
Composition: EPS Group, Inc., Hanover, Maryland
Set in: Times Roman
Printing: Victor Graphics, Inc., Baltimore, Maryland
Binding: American Trade Bindery, Baltimore, Maryland
Cover Design: DR Pollard and Associates, Inc., Arlington, Virginia
Color in water may result from the presence of natural metallic ions (iron and manganese), humus and peat materials, plankton, weeds, and industrial wastes. Color is removed to make a water suitable for general and industrial applications. Colored industrial wastewaters may require color removal before discharge into watercourses.
The term “color” is used here to mean true color, that is, the color of water from which turbidity has been removed. The term “apparent color” includes not only color due to substances in solution, but also that due to suspended matter. Apparent color is determined on the original sample without filtration or centrifugation. In some highly colored industrial wastewaters color is contributed principally by colloidal or suspended material. In such cases both true color and apparent color should be determined.
* Approved by Standard Methods Committee. 1988.
2-1To determine color by currently accepted methods, turbidity must be removed before analysis. The optimal method for removing turbidity without removing color has not been found yet. Filtration yields results that are reproducible from day to day and among laboratories. However, some filtration procedures also may remove some true color. Centrifugation avoids interaction of color with filter materials, but results vary with the sample nature and size and speed of the centrifuge. When sample dilution is necessary, whether it precedes or follows turbidity removal, it can alter the measured color if large color-bodies are present.
Acceptable pretreatment procedures are included with each method. State the pretreatment method when reporting results.
The visual comparison method is applicable to nearly all samples of potable water. Pollution by certain industrial wastes may produce unusual colors that cannot be matched. In this case use an instrumental method. A modification of the tristimulus and the spectrophotometric methods allows calculation of a single color value representing uniform chromaticity differences even when the sample exhibits color significantly different from that of platinum cobalt standards. For comparison of color values among laboratories, calibrate the visual method by the instrumental procedures.
OPTICAL SOCIETY OF AMERICA. 1943. Committee Report. The concept of color. J. Opt. Soc. Amer. 33:544.
JONES, H. et al. 1952. The Science of Color. Thomas Y. Crowell Co., New York, N. Y.
a. Principle: Color is determined by visual comparison of the sample with known concentrations of colored solutions. Comparison also may be made with special, properly calibrated glass color disks. The platinum-cobalt method of measuring color is the standard method, the unit of color being that produced by 1 mg platinum/L in the form of the chloroplatinate ion. The ratio of cobalt to platinum may be varied to match the hue in special cases; the proportion given below is usually satisfactory to match the color of natural waters.
b. Application: The platinum-cobalt method is useful for measuring color of potable water and of water in which color is due to naturally occurring materials. It is not applicable to most highly colored industrial wastewaters.
c. Interference: Even a slight turbidity causes the apparent color to be noticeably higher than the true color; therefore remove turbidity before approximating true color by differential reading with different color filters1 or by differential scattering measurements.2 Neither technique, however, has reached the status of a standard method. Remove turbidity by centrifugation or by the filtration procedure described under Method C. Centrifuge for 1 h unless it has been demonstrated that centrifugation under other conditions accomplishes satisfactory turbidity removal.
The color value of water is extremely pH-dependent and invariably increases as the pH of the water is raised. When reporting a color value, specify the pH at which color is determined. For research purposes or when color values are to be compared among laboratories, determine the color response of a given water over a wide range of pH values.3
d. Field method: Because the platinum-cobalt standard method is not convenient for field use, compare water color with that of glass disks held at the end of metallic tubes containing glass comparator tubes filled with sample and colorless distilled water. Match sample color with the color of the tube of clear water plus the calibrated colored glass when viewed by looking toward a white surface. Calibrate each disk to correspond with the colors on the platinum-cobalt scale. The glass disks give results in substantial agreement with those obtained by the platinum-cobalt method and their use is recognized as a standard field procedure.
e. Nonstandard laboratory methods: Using glass disks or liquids other than water as standards for laboratory work is permissible only if these have been individually calibrated against platinum-cobalt standards. Waters of highly unusual color, such as those that may occur by mixture with certain industrial wastes, may have hues so far removed from those of the platinum-cobalt standards that comparison by the standard method is difficult or impossible. For such waters, use the methods in Sections 2120C and D. However, results so obtained are not directly comparable to those obtained with platinum-cobalt standards.
f. Sampling: Collect representative samples in clean glassware. Make the color determination within a reasonable period because biological or physical changes occurring in storage may affect color. With naturally colored waters these changes invariably lead to poor results.
a. Nessler tubes, matched, 50-mL, tall form.
b. pH meter, for determining sample pH (see Section 4500–H+).
a. If a reliable supply of potassium chloroplatinate cannot be purchased, use chloroplatinic acid prepared from metallic platinum. Do not use commercial chloroplatinic acid because it is very hygroscopic and may vary in platinum content. Potassium chloroplatinate is not hygroscopic.
b. Dissolve 1.246 g potassium chloroplatinate, K2PtCl6 (equivalent to 500 mg metallic Pt) and 1.00 g crystallized cobaltous chloride, CoCl26H2O (equivalent to about 250 mg metallic Co) in distilled water with 100 mL conc HCl and dilute to 1000 mL with distilled water. This stock standard has a color of 500 units.
2-2c. If K2PtCl6 is not available, dissolve 500 mg pure metallic Pt in aqua regia with the aid of heat; remove HNO3 by repeated evaporation with fresh portions of conc HCl. Dissolve this product, together with 1.00 g crystallized CoCl2·6H2O, as directed above.
d. Prepare standards having colors of 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 60, and 70 by diluting 0.5, 1.0, 1.5, 2.0, 2.5, 3.0, 3.5, 4.0, 4.5, 5.0, 6.0, and 7.0 mL stock color standard with distilled water to 50 mL in nessler tubes. Protect these standards against evaporation and contamination when not in use.
a. Estimation of intact sample: Observe sample color by filling a matched nessler tube to the 50-mL mark with sample and comparing it with standards. Look vertically downward through tubes toward a white or specular surface placed at such an angle that light is reflected upward through the columns of liquid. If turbidity is present and has not been removed, report as “apparent color.” If the color exceeds 70 units, dilute sample with distilled water in known proportions until the color is within the range of the standards.
b. Measure pH of each sample.
a. Calculate color units by the following equation:
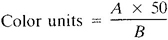
where:
A = estimated color of a diluted sample and
B = mL sample taken for dilution.
b. Report color results in whole numbers and record as follows:
| Color Units | Record to Nearest |
| 1—50 | 1 |
| 51—100 | 5 |
| 101—250 | 10 |
| 251—500 | 20 |
c. Report sample pH.
HAZEN, A. 1892. A new color standard for natural waters. Amer. Chem. J. 14:300.
HAZEN, A. 1896. The measurement of the colors of natural waters. J. Amer. Chem. Soc. 18:264.
Measurement of Color and Turbidity in Water. 1902. U.S. Geol. Surv., Div. Hydrog. Circ. 8, Washington, D.C.
RUDOLES, W. & W.D. HANLON. 1951. Color in industrial wastes. Sewage Ind. Wastes 23:1125.
PALIN, A. T. 1955. Photometric determination of the colour and turbidity of water. Water Water Eng. 59:341.
CHRISTMAN, R.F. & M. GHASSEMI. 1966. Chemical nature of organic color in water. J. Amer. Water Works Assoc. 58:723.
GHASSEMI, M. & R.F. CHRISTMAN, 1968. Properties of the yellow organic acids of natural waters. Limnol. Oceanogr. 13:583.
a. Principle: The color of a filtered sample is expressed in terms that describe the sensation realized when viewing the sample. The hue (red, green, yellow, etc.) is designated by the term “dominant wavelength,” the degree of brightness by “luminance,” and the saturation (pale, pastel, etc.) by “purity.” These values are best determined from the light transmission characteristics of the filtered sample by means of a spectrophotometer.
b. Application: This method is applicable to potable and surface waters and to wastewaters, both domestic and industrial.
c. Interference: Turbidity interferes. Remove by the filtration method described below.
a. Spectrophotometer, having 10-mm absorption cells, a narrow (10-nm or less) spectral band, and an effective operating range from 400 to 700 nm.
b. Filtration system, consisting of the following (see Figure 2120:1):
1) Filtration flasks, 250-mL, with side tubes.
2) Walter crucible holder.
3) Micrometallic filter crucible, average pore size 40 µm.
4) Calcined filter aid.*
5) Vacuum system.
a. Preparation of sample: Bring two 50-mL samples to room temperature. Use one sample at the original pH; adjust pH of the other to 7.6 by using sulfuric acid (H2SO4) and sodium hydroxide (NaOH) of such concentrations that the resulting volume change does not exceed 3%. A standard pH is necessary because of the variation of colour with pH. Remove excessive quantities
* Celite No. 505, Manville Corp., or equivalent.
2-3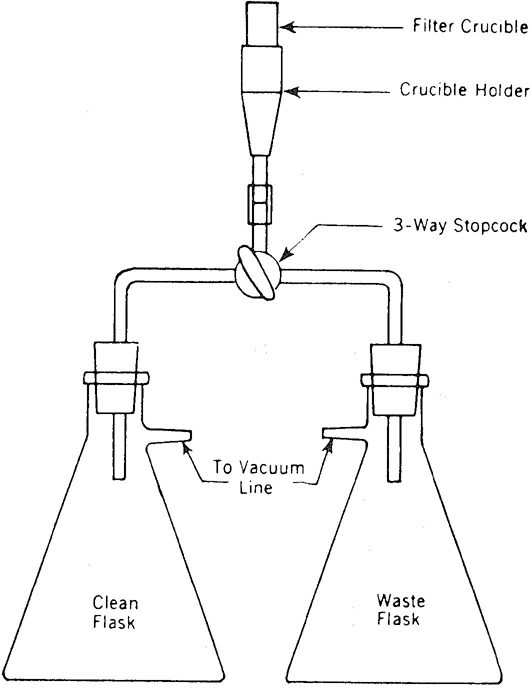
Figure 2120:1. Filtration system for colour determinations.
of suspended materials by centrifuging. Treat each sample separately, as follows:
Thoroughly mix 0.1 g filter aid in a 10-mL portion of centrifuged sample and filter to form a precoat in the filter crucible. Direct filtrate to waste flask as indicated in Figure 2120:1. Mix 40 mg filter aid in a 35-mL portion of centrifuged sample. With vacuum still on, filter through the precoat and pass filtrate to waste flask until clear; then direct clear-filtrate flow to clean flask by means of the three-way stopcock and collect 25 mL for the transmittance determination.
b. Determination of light transmission characteristics: Thoroughly clean 1-cm absorption cells with detergent and rinse with distilled water. Rinse twice with filtered sample, clean external surfaces with lens paper, and fill cell with filtered sample.
Determine transmittance values (in percent) at each visible wavelength value presented in Table 2120:1, using the 10 ordinates marked with an asterisk for fairly accurate work and all 30 ordinates for increased accuracy. Set instrument to read 100% transmittance on the distilled water blank and make all determinations with a narrow spectral band.
a. Tabulate transmittance values corresponding to wavelengths shown in Columns X, Y, and Z in Table 2120:I. Total each transmittance column and multiply totals by the appropriate factors (for 10 or 30 ordinates) shown at the bottom of the table, to obtain tristimulus values X, Y, and Z. The tristimulus value Y is percent luminance.
| Ordinate No. | X | Y | Z |
|---|---|---|---|
| Wavelength nm |
|||
| 1 | 424.4 | 465.9 | 414.1 |
| 2* | 435.5* | 489.5* | 422.2* |
| 3 | 443.9 | 500.4 | 426.3 |
| 4 | 452.1 | 508.7 | 429.4 |
| 5* | 461.2* | 515.2* | 432.0* |
| 6 | 474.0 | 520.6 | 434.3 |
| 7 | 531.2 | 525.4 | 436.5 |
| 8* | 544.3* | 529.8* | 438.6* |
| 9 | 552.4 | 533.9 | 440.6 |
| 10 | 558.7 | 537.7 | 442.5 |
| 11* | 564.1* | 541.4* | 444.4* |
| 12 | 568.9 | 544.9 | 446.3 |
| 13 | 573.2 | 548.4 | 448.2 |
| 14* | 577.4* | 551.8* | 450.1* |
| 15 | 581.3 | 555.1 | 452.1 |
| 16 | 585.0 | 558.5 | 454.0 |
| 17* | 588.7* | 561.9* | 455.9* |
| 18 | 592.4 | 565.3 | 457.9 |
| 19 | 596.0 | 568.9 | 459.9 |
| 20* | 599.6* | 572.5* | 462.0* |
| 21 | 603.3 | 576.4 | 464.1 |
| 22 | 607.0 | 580.4 | 466.3 |
| 23* | 610.9* | 584.8* | 468.7* |
| 24 | 615.0 | 589.6 | 471.4 |
| 25 | 619.4 | 594.8 | 474.3 |
| 26* | 624.2* | 600.8* | 477.7* |
| 27 | 629.8 | 607.7 | 481.8 |
| 28 | 636.6 | 616.1 | 487.2 |
| 29* | 645.9* | 627.3* | 495.2* |
| 30 | 663.0 | 647.4 | 511.2 |
| Factors when 30 Ordinates Used | |||
| 0.032.69 | 0.033.33 | 0.039.38 | |
| Factors When 10 Ordinates Used | |||
| 0.098.06 | 0.100.00 | 0.118.14 | |
| * Insert in each column the transmittance value (%) corresponding to the wavelength shown. Where limited accuracy is sufficient, use only the ordinates marked with an asterisk. | |||
b. Calculate the trichromatic coefficients x and y from the tristimulus values X, Y, and Z by the following equations:
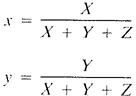
Locate point (x, y) on one of the chromaticity diagrams in Figure 2120:2 and determine the dominant wavelength (in nanometers) and the purity (in percent) directly from the diagram.
2-4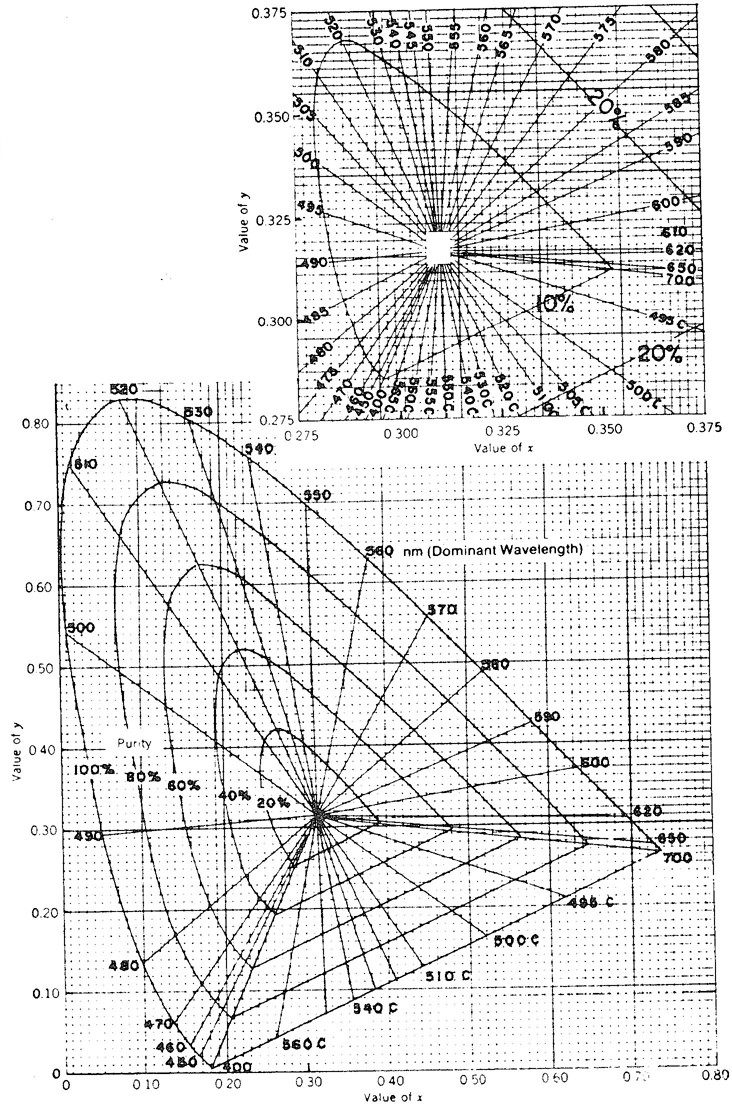
Figure 2120:2. Chromaticity diagrams.
2-5| Wavelength Range nm |
Hue |
|---|---|
| 400–465 | Violet |
| 465–482 | Blue |
| 482–497 | Blue–green |
| 497–530 | Green |
| 530–575 | Greenish yellow |
| 575–580 | Yellow |
| 580–587 | Yellowish orange |
| 587–598 | Orange |
| 598–620 | Orange–red |
| 620–700 | Red |
| 400–530c* | Blue–purple |
| 530c–700* | Red–purple |
| * See Figure 2120:2 for significance of “c”. | |
Determine hue from the dominant-wavelength value, according to the ranges in Table 2120:II.
Express color characteristics (at pH 7.6 and at the original pH) in terms of dominant wavelength (nanometers, to the nearest unit), hue (e.g., blue, blue-green, etc.) luminance (percent, to the nearest tenth), and purity (percent, to the nearest unit). Report type of instrument (i.e., spectrophotometer), number of selected ordinates (10 or 30), and the spectral band width (nanometers) used.
HARDY, A.C. 1936. Handbook of Colorimetry. Technology Press, Boston, Mass.
a. Principle: Three special tristimulus light filters, combined with a specific light source and photoelectric cell in a filter photometer, may be used to obtain color data suitable for routine control purposes.
The percentage of tristimulus light transmitted by the solution is determined for each of the three filters. The transmittance values then are converted to trichromatic coefficients and color characteristic values.
b. Application: This method is applicable to potable and surface waters and to wastewaters, both domestic and industrial. Except for most exacting work, this method gives results very similar to the more accurate Method C.
c. Interference: Turbidity must be removed.
a. Filter photometer.*
b. Filter photometer light source: Tungsten lamp at a color temperature of 3000°C.†
c. Filter photometer photoelectric cells, 1 cm.‡
d. Tristimulus filters.§
e. Filtration system: See Section 2120C.2b and Figure 2120:1.
a. Preparation of sample: See Section 2120C.3a.
b. Determination of light transmission characteristics: Thoroughly clean (with detergent) and rinse 1-cm absorption cells with distilled water. Rinse each absorption cell twice with filtered sample, clean external surfaces with lens paper, and fill cell with filtered sample.
Place a distilled water blank in another cell and use it to set the instrument at 100% transmittance. Determine percentage of light transmission through sample for each of the three tristimulus light filters, with the filter photometer lamp intensity switch in a position equivalent to 4 V on the lamp.
a. Determine luminance value directly as the percentage transmittance value obtained with the No. 2 tristimulus filter.
b. Calculate tristimulus values X, Y, and Z from the percentage transmittance (T1, T2, T3) for filters No. 1, 2, 3, as follows:
X = T3 × 0.06 + T1 × 0.25
Y = T2 × 0.316
Z = T3 × 0.374
Calculate and determine trichromatic coefficients x and y, dominant wavelength, hue, and purity as in Section 2120C.4b above.
Express results as prescribed in Section 2120C.5.
* Fisher Electrophotometer or equivalent.
† General Electric lamp No. 1719 (at 6 V) or equivalent.
‡ General Electric photovoltaic cell. Type PV-1, or equivalent.
§ Corning CS-3-107 (No. 1), CS-4-98 (No. 2), and CS-5-70 (No. 3), or equivalent.
2-6a. Principle: This method is an extension of Tristimulus Method 2120D. By this method a measure of the sample color, independent of hue, may be obtained. It is based on use of the Adams-Nickerson chromatic value formula1 for calculating single number color difference values. i.e., uniform color differences. For example, if two colors, A and B, are judged visually to differ from colorless to the same degree, their ADMI color values will be the same. The modification was developed by members of the American Dye Manufacturers Institute (ADMI).2
b. Application: This method is applicable to colored waters and wastewaters having color characteristics significantly different from platinum-cobalt standards, as well as to waters and wastewaters similar in hue to the standards.
c. Interference: Turbidity must be removed.
a. Filter photometer* equipped with CIE tristimulus filters (see 2120D. 2d).
b. Filter photometer light source: Tungsten lamp at a color temperature of 3000°C (see 2120D.2b).
c. Absorption cells and appropriate cell holders: For color values less than 250 ADMI units, use cells with a 5.0-cm light path; for color values greater than 250, use cells with 1.0-cm light path.
d. Filtration system: See Section 2120C.2b and Figure 2120:1; or a centrifuge capable of achieving 1000 × g. (See Section 2120B.)
a. Instrument calibration: Establish curves for each photometer; calibration data for one instrument cannot be applied to another one. Prepare a separate calibration curve for each absorption cell path length.
1) Prepare standards as described in 2120B.3. For a 5-cm cell length prepare standards having color values of 25, 50, 100, 200, and 250 by diluting 5.0, 10.0, 20.0, 30.0, 40.0, and 50.0 mL stock color standard with distilled water to 100 mL in volumetric flasks. For the shorter pathlength, prepare appropriate standards with higher color values.
2) Determine light transmittance (see ¶ 3c, below) for each standard with each filter.
3) Using the calculations described in ¶ 3d below, calculate the tristimulus values (Xs, Ys, Zs) for each standard, determine the Munsell values, and calculate the intermediate value (DE).
4) Using the DE values for each standard, calculate a calibration factor Fn for each standard from the following equation:
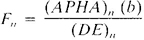
where:
(APHA)n, = APHA color value for standard n.
(DE)n = intermediate value calculated for standard n, and
b = cell light path, cm.
Placing (DE)n on the X axis and Fn on the Y axis, plot a curve for the standard solutions. Use calibration curve to derive the F value from DE values obtained with samples.
b. Sample preparation: Prepare two 100-mL sample portions (one at the original pH. one at pH 7.6) as described in Section 2120C.3a, or by centrifugation. (NOTE: Centrifugation is acceptable only if turbidity removal equivalent to filtration is achieved.)
c. Determination of light transmission characteristics: Thoroughly clean absorption cells with detergent and rinse with distilled water. Rinse each absorption cell twice with filtered sample. Clean external surfaces with lens paper and fill cell with sample. Determine sample light transmittance with the three filters to obtain the transmittance values: T1 from Filter 1. T2 from Filter 2, and T3 from Filter 3. Standardize the instrument with each filter at 100% transmittance with distilled water.
d. Calculation of color values: Tristimulus values for samples are Xs, Ys, and Zs; for standards Xr, Yr, and Zr, and for distilled water Xc, Yc, and Zc. Munsell values for samples are Vxs, Vys and Vzs; for standards, Vxr, Vyr, and Vzr, and for distilled water Vxc, Yyc, and Vzc.
For each standard or sample calculate the tristimulus values from the following equations:
X = (T3 × 0.1899) + (T1 × 0.791)
Y = T2
Z = T3 × 1.1835
Tristimulus values for the distilled water blank used to standardize the instrument are always:
Xc = 98.09
Yc = 100.0
Zc = 118.35
Convert the six tristimulus values (Xs, Ys, Zs Xc, Yc, Zc) to the corresponding Munsell values using published tables 2, 3, 4† or by the equation given by Bridgeman.3
Calculate the intermediate value of DE from the equation:
DE = {(0.23 ΔVy)2 + [Δ(Vx − Vy)]2 + [0.4 Δ(Vy − Vz)2}½
where:
Vy = Vys − Vyc
Δ(VA − Vv) = (Vxs − Vys) − (Vxc − Vvc)
Δ(Vy − Vz) = (Vvs − Vzs) − (Vvc − Vzc)
when the sample is compared to distilled water.
* Fisher Electrocolorimeter. Model 181, or equivalent.
† Instrumental Colour Systems. Ltd. 7 Bucklebury Place. Upper Woolhampton, Berkshire RG7 5UD, England.
2-7With the standard calibration curve, use the DE value to determine the calibration factor F.
Calculate the final ADMI color value as follows:
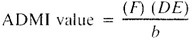
where:
b = absorption cell light path, cm.
Report ADMI color values at pH 7.6 and at the original pH.
The ADMI color value also may be determined spectrophotometrically, using a spectrophotometer with a narrow (10-nm or less) spectral band and an effective operating range of 400 to 700 nm. This method is an extension of 2120C. Tristimulus values may be calculated from transmittance measurements, preferably by using the weighted ordinate method or by the selected ordinate method. The method has been described by Allen et al.,2 who include work sheets and worked examples.
JUDD. D.B. & G. WYSZECKI. 1963. Color in Business, Science, and Industry, 2nd ed. John Wiley & Sons, New York, N.Y. (See Tables A, B, and C in Appendix.)
WYSZECKI. G. & W.S. STILES. 1967. Color Science. John Wiley & Sons. New York, N.Y. (See Tables 6.4. A, B, C, pp. 462–467.)
2-8